 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的 =-30.6kJ·mol
Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的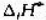 =-30.6kJ·mol-1,
=-30.6kJ·mol-1,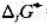 =-11.2kJ·mol-1,试求在298K时
=-11.2kJ·mol-1,试求在298K时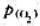 的压力是多少(Pa)?Ag2O(s)的热分解温度是多少?(105Pa下)
的压力是多少(Pa)?Ag2O(s)的热分解温度是多少?(105Pa下)
 答案
答案
 请输入或粘贴题目内容
搜题
请输入或粘贴题目内容
搜题
 拍照、语音搜题,请扫码下载APP
拍照、语音搜题,请扫码下载APP
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的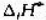 =-30.6kJ·mol-1,
=-30.6kJ·mol-1,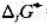 =-11.2kJ·mol-1,试求在298K时
=-11.2kJ·mol-1,试求在298K时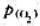 的压力是多少(Pa)?Ag2O(s)的热分解温度是多少?(105Pa下)
的压力是多少(Pa)?Ag2O(s)的热分解温度是多少?(105Pa下)
 答案
答案
 更多“Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的 =-30.6kJ·mol”相关的问题
更多“Ag2O遇热分解:2Ag2O(s)=4Ag(s)+O2(g),已知在298K时,Ag2O(s)的 =-30.6kJ·mol”相关的问题
第1题
已知300K时: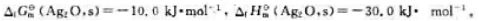
(1)试通过计算说明在300K、标准状态下、Ag2O(s)能否发生如下分解反应:
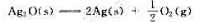
(2)计算在标准状态下Ag2O(s)发生分解反应的最低温度。
第3题
Ag2CO3遇热分解
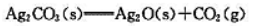
383K时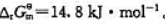 383K烘干时,空气中加入多少CO2可避免Ag2CO3分解?
383K烘干时,空气中加入多少CO2可避免Ag2CO3分解?
第4题
Silver oxide,Ag2O,can decompose into its elements,Ag(S)and O2(g).(a)Write a balanced equation for this decomposition reaction.(b)Calculate the
第6题
在67℃时,获得N2O5(g)分解反应
| t/s | 0.0 | 60.0 | 120.0 | 180.0 | 240.0 |
| c(N2O5)/(mol·L-1) | 0.160 | 0.113 | 0.080 | 0.056 | 0.040 |
第8题
A.三角分解的命令格式为:[l,u]=lu(A)
B.求A的转置矩阵的命令格式为:conj'(A)
C.将矩阵A扩展为4列的命令格式为:A(:,4)=[5;4;3]
D.奇异值分解的命令格式为:[u,s,v]=svd(A)
第10题
第11题
如图所示,当US=10V,IS=2A时,电流I=4A;当US=5V,IS=4A时,I=6A。求当US=15V,IS=3A时,电流I是多少?
