题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
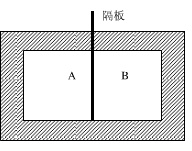
一个刚性容器被绝热隔板分成A和B两部分(如图5-5所示)。已知A和B中的状态分别为:mA=1kg,xA=1,pA=0.5MPa;mB=2k
一个刚性容器被绝热隔板分成A和B两部分(如图5-5所示)。已知A和B中的状态分别为:mA=1kg,xA=1,pA=0.5MPa;mB=2kg,xB=0.8,pB=1MPa,假定抽去隔板后容器内的最终压力为0.7MPa,若环境温度为300K,试求:

 答案
答案
刚性容器的容积及蒸汽的最终状态
态A是干饱和蒸汽,根据压力0.5MPa可由附表11查出
V'A=0.38905m3/kg,h"A=2747.57kJ/kg,s"A=6.82805kJ/(kg·K)
VA=mAv'A=1×0.38905=0.38905m3
态B是xB=0.8的湿蒸汽,根据压力1MPa可由附表11查出
v'=0.0011272,v"=0.19438m3/kg,h'=762.84,h"=2777.67kJ/kg
s'=2.1388,s"=6.5859kJ/(kg·K)
vx=xv"+(1-x)v'=0.8×0.19438+0.2×0.0011272=0.15573m3/kg
VB=mBvx=2×0.15573=0.3114m3
hB=xh"+(1-x)h'=0.8×2777.67+0.2×762.84=2374.7kJ/kg
sB=xs"+(1-x)s'=0.8×6.5859+0.2×2.1388=5.6965kJ/(kg·K)
终态的容积V=0.38905+0.3114=0.70044m3,质量m=3kg,因此有
v=V/m=0.70044/3=0.23348m3/kg
根据压力0.7MPa可由附表11查出
h'=697.32,h"=2763.29kJ/kg
s'=1.9925,s"=6.7079kJ/(kg·K)
v'=0.0011079,v"=0.27281>v=0.23348m3/kg
x=(0.23348-0.0011079)/(0.27281-0.0011079)=0.855
hx=xh"+(1-x)h'=0.855×2763.29+0.145×697.32=2463.7kJ/kg
sx=xs"+(1-x)s'=0.855×6.7079+0.145×1.9925=6.024kJ/(kg·K)$过程中交换的热量
对整体写出能量方程,有Q=U-(UA+UB),其中
U=H-pV=3×2463.7-700×0.70044=6900.8kJ
UA=mAh"A-pAVA=1×2747.57-500×0.38905=2553.04kJ
UB=mBhB-pBVB=2×2374.7-1000×0.3114=4438kJ
Q=6900.8-(2553.04+4438)=-90.2kJ=-Q0$过程中总的熵产
根据孤立系统的熵方程,不难得出
SPtot=ΔS+ΔS0=ΔS+Q0/T0
=3×6.024-(1×6.82805+2×5.6965)+90.2/300=0.1516kJ/K