 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
The [Ag+] of a solution is 4.0×10-5. Calculate the [Cl-] that must be exceeded before AgCl can preci
The [Ag+] of a solution is 4.0×10-5. Calculate the [Cl-] that must be exceeded before AgCl can precipitate.
 答案
答案
 请输入或粘贴题目内容
搜题
请输入或粘贴题目内容
搜题
 拍照、语音搜题,请扫码下载APP
拍照、语音搜题,请扫码下载APP
 题目内容
(请给出正确答案)
题目内容
(请给出正确答案)
The [Ag+] of a solution is 4.0×10-5. Calculate the [Cl-] that must be exceeded before AgCl can precipitate.
 答案
答案
 更多“The [Ag+] of a solution is 4.0×10-5. Calculate the [Cl-] that must be exceeded before AgCl can preci”相关的问题
更多“The [Ag+] of a solution is 4.0×10-5. Calculate the [Cl-] that must be exceeded before AgCl can preci”相关的问题
第1题
The world “countermeasures” in paragraph 2 means
A.“problems”
B.“approaches”
C.“solutions”
D.“actions”
第2题
听录音,回答以下问题:
A.The cause of the health care problem in the US.
B.The solution to the health care problem in the US.
C.The condition of the health care problem in the US.
D.The reform. of the health care system in the US.
第4题
已知

 逆向自发进行。 ( )
逆向自发进行。 ( )
第7题
298K时,Ag(s)|Ag+(a)的标准电极电势EΘ=0.799V,反应Fe3+(a2)+Ag(s)→Fe2+(a3)+Ag+(a1)的平衡常数KΘ=0.531,试求
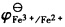 。
。